 
The law of conservation of mass states that the total mass present before a chemical reaction is the same as the total mass present after the chemical reaction. Atomic theory is the collection of ideas that have morphed over time as more and more information about the structure of the atom has become clearer. What is Atomic Theory Dalton’s atomic theory was a scientific theory on the nature of matter put forward by the English physicist and chemist John Dalton in the year 1808. The notion that the building blocks of matter are invisibly tiny particles called atoms is usually traced back to the Greek philosophers Leucippus of Miletus and Democritus of Abdera in the 5 th Century BC. Every solid, liquid, gas, and plasma consists of neutral or ionized atoms. 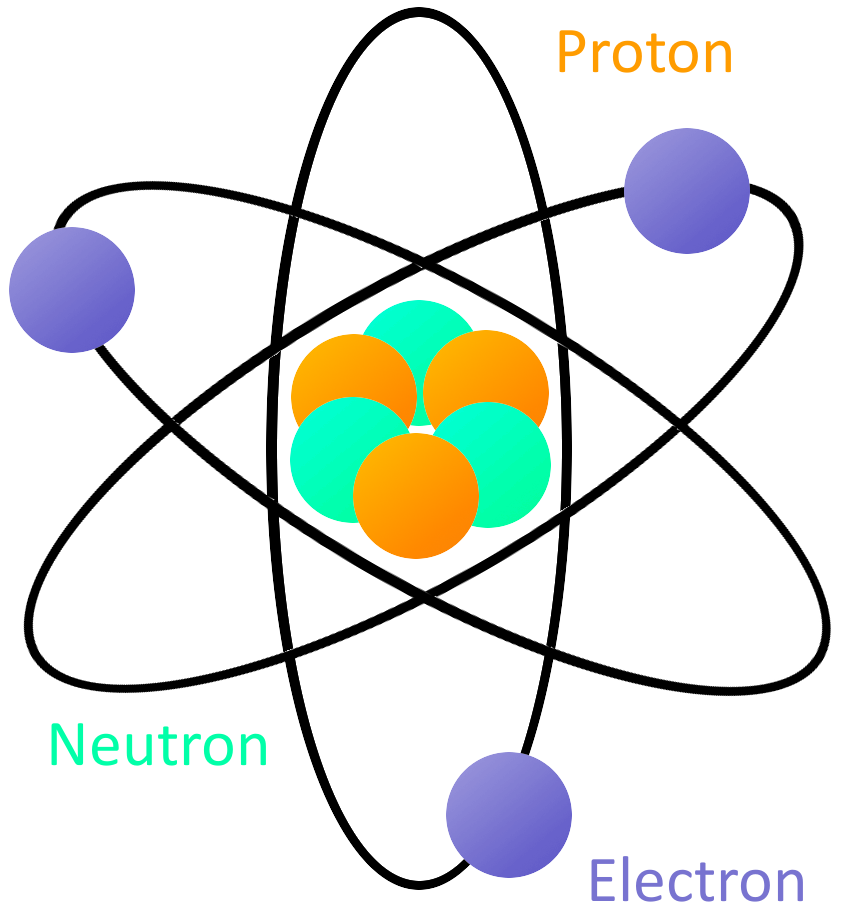
An atom is the smallest unit of matter that forms a chemical element and cannot be divided using any chemical means. In the fields of quantum mechanics and atomic theory, these mathematical functions are often employed in order to determine the probability of finding an electron (belonging to an atom) in a specific region around the nucleus of the atom. The concept that matter is composed of discrete particles is an ancient idea, but gained scientific credence in the 18th and 19th centuries when scientists found it could explain the behaviors of gases and how chemical elements reacted with each other. Rutherford model, description of the structure of atoms proposed (1911) by the New Zealand-born physicist Ernest Rutherford. An atom is the basic building block of matter, consisting of protons, neutrons, and electrons. Scientists did not account for the gases that play a critical role in this reaction. The atomic theory led to the creation of the law of multiple proportions. Atomic theory is the scientific theory that matter is composed of particles called atoms. Thomson atomic model, earliest theoretical description of the inner structure of atoms, proposed about 1900 by William Thomson (Lord Kelvin) and strongly supported by Sir Joseph John Thomson, who had discovered (1897) the electron, a negatively charged part of every atom. Atomic theory is a scientific description of the nature of atoms and matter that combines elements of physics, chemistry, and mathematics. A theory of chemical combination, first stated by John Dalton in 1803. 
Thomson produced a visible beam in a cathode ray tube.\) shows that the burning of word does follow the law of conservation of mass. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
